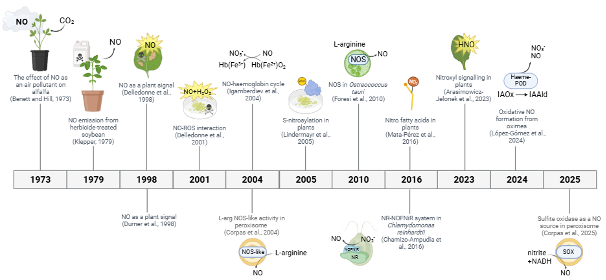
Brief History of Plant NO
Early studies on nitric oxide (NO) in plants began with nitrate reduction research in the 1960s, showing NO involvement in various crops (Fewson and Nicholas 1960). In the 1970s and 1980s, work on air pollution and herbicide responses in species like alfalfa and soybean suggested NO played physiological roles (Bennett and Hill, 1973; Klepper 1979). The field gained momentum in 1998 with studies demonstrating NO as a signalling molecule in plant-pathogen interactions, similar to its role in animals (Durner et al., 1998; Delledonne et al., 1998).
Subsequent research found NO production in various plant systems, often linked to a NOS-like enzyme, despite the absence of a confirmed NOS gene in higher plants. In 1999, peroxisomes (Barroso et al., 1999) and purified nitrate reductase (NR, Yamasaki et al., 1999) were shown to generate NO.
In 2001, it was shown that NO interacts with reactive oxygen species (ROS) during pathogen responses (Delledonne et al., 2001). In 2004, the haemoglobin-NO cycle was proposed to explain NO regulation during hypoxia (Igamberdiev et al., 2004).
Key discoveries in 2005–2006 included NO generation by mitochondria (Planchet et al., 2005) and chloroplasts (Jasid et al., 2006), as well as protein S-nitrosation as a major signalling mechanism in plants (Lindermayr et al., 2005). Meanwhile, plant S-nitrosoglutathione reductase (GSNOR) was reported (Feechan et al., 2005) using plant disease resistance as a basis for the work. This showed that there was a mechanism able to reverse thiol-based covalent changes induced by NO accumulation in cells, showing how signalling in plants cells could be regulated in plants. In 2006, regulation of NO via the haemoglobin-NO cycle was further elaborated (Igamberdiev et al., 2006).
Despite efforts, a true plant NOS has not been identified, though a NOS enzyme was discovered in the alga Ostreococcus tauri in 2010 (Foresi et al., 2010). Later studies (2016) introduced the concept of NO signalling via fatty acid modifications (Mata-Pérez et al., 2016) and described the NR-NOFNiR system for NO production in Chlamydomonas (Chamizo-Ampudia et al., 2016).
More recently, in 2023, nitroxyl (HNO) was proposed as a reactive signalling molecule (Arasimowicz-Jelonek et al., 2023), and in 2024, oxidative NO generation from oximes was reported (López-Gómez et al., 2024), adding further complexity to NO biology in plants. In 2025, sulfite oxidase as a new player in plant NO synthesis has been described in plants (Corpas et al., 2025).
A timeline of the most significant reports in plant research is shown in the Figure (Hancock et al., 2025).
References
- Arasimowicz-Jelonek, M., Floryszak-Wieczorek, J., Suarez, S., Doctorovich, F., Sobieszczuk-Nowicka, E., Bruce King, S., Milczarek, G., Rębiś, T., Gajewska, J., Jagodzik, P. and Żywicki, M. Discovery of endogenous nitroxyl as a new redox player in Arabidopsis thaliana. Nature Plants 9, 36-44 (2023).
- Barroso, J.B., Corpas, F.J., Carreras, A., Sandalio, L.M., Valderrama, R., Palma, J., Lupiánez, J.A. and del Rıo, L.A. Localization of nitric-oxide synthase in plant peroxisomes. Journal of Biological Chemistry 274, 36729-36733 (1999).
- Bennett, J.H. and Hill, A.C. Inhibition of apparent photosynthesis by air pollutants. American Society of Agronomy, Crop Science Society of America, and Soil Science Society of America 2, 526-530 (1973).
- Chamizo‐Ampudia, A., Sanz‐Luque, E., Llamas, Á., Ocaña‐Calahorro, F., Mariscal, V., Carreras, A., Barroso, J.B., Galván, A. and Fernández, E. A dual system formed by the ARC and NR molybdoenzymes mediates nitrite‐dependent NO production in Chlamydomonas. Plant, Cell & Environment 39, 2097-2107 (2016).
- Corpas, F.J., Taboada, J., Sánchez-Romera, B., López-Jaramillo, J. and Palma, J.M. Peroxisomal Sulfite Oxidase (SOX), an alternative source of NO in higher plants which is upregulated by H2S. Plant Physiology and Biochemistry, 110000 (2025).
- Delledonne, M., Xia, Y., Dixon, R.A. and Lamb, C. Nitric oxide functions as a signal in plant disease resistance. Nature 394, 585-588 (1998).
- Delledonne, M., Zeier, J., Marocco, A. and Lamb, C. Signal interactions between nitric oxide and reactive oxygen intermediates in the plant hypersensitive disease resistance response. Proceedings of the National Academy of Sciences, USA 98, 13454-13459 (2001).
- Durner, J., Wendehenne, D. and Klessig, D.F. Defense gene induction in tobacco by nitric oxide, cyclic GMP, and cyclic ADP-ribose. Proceedings of the National Academy of Sciences, USA 95, 10328-10333 (1998).
- Feechan, A., Kwon, E., Yun, B.W., Wang, Y., Pallas, J.A. and Loake, G.J. A central role for S-nitrosothiols in plant disease resistance. Proceedings of the National Academy of Sciences, USA 102, 8054-8059 (2005).
- Fewson, C.A. and Nicholas, D.J. Utilization of nitric oxide by micro-organisms and higher plants. Nature 188, 794-6 (1960).
- Foresi, N., Correa-Aragunde, N., Parisi, G., Calo, G., Salerno, G. and Lamattina, L. Characterization of a nitric oxide synthase from the plant kingdom: NO generation from the green alga Ostreococcus tauri is light irradiance and growth phase dependent. The Plant Cell 22, 3816-3830 (2010).
- Hancock, J.T., Corpas, F.J., Silveira, N.M., Kolbert, Zs. A brief history of nitric oxide in plants: How we got here, where we are, and where we might be going. Journal of Plant Physiology and Metabolism (2025).
- Igamberdiev, A.U. and Hill, R.D. Nitrate, NO and haemoglobin in plant adaptation to hypoxia: an alternative to classic fermentation pathways. Journal of Experimental Botany 55, 2473-2482 (2004).
- Igamberdiev, A.U., Stoimenova, M., Seregélyes, C. and Hill, R.D. Class-1 hemoglobin and antioxidant metabolism in alfalfa roots. Planta 223, 1041-1046 (2006).
- Jasid, S., Simontacchi, M., Bartoli, C.G. and Puntarulo, S. Chloroplasts as a nitric oxide cellular source. Effect of reactive nitrogen species on chloroplastic lipids and proteins. Plant Physiology 142, 1246-1255 (2006).
- Klepper, L. Nitric oxide (NO) and nitrogen dioxide (NO2) emissions from herbicide-treated soybean plants. Atmospheric Environment (1967) 13, 537-542 (1979).
- Lindermayr, C., Saalbach, G. and Durner, J. Proteomic identification of S-nitrosylated proteins in Arabidopsis. Plant Physiology 137, 921-930 (2005).
- López-Gómez, P., Buezo, J., Urra, M., Cornejo, A., Esteban, R., de Los Reyes, J.F., Urarte, E., Rodríguez-Dobreva, E., Chamizo-Ampudia, A., Eguaras, A. and Wolf, S. A new oxidative pathway of nitric oxide production from oximes in plants. Molecular Plant 17, 178-198 (2024).
- Mata-Pérez, C., Sánchez-Calvo, B., Padilla, M.N., Begara-Morales, J.C., Luque, F., Melguizo, M., Jiménez-Ruiz, J., Fierro-Risco, J., Peñas-Sanjuán, A., Valderrama, R. and Corpas, F.J. Nitro-fatty acids in plant signaling: nitro-linolenic acid induces the molecular chaperone network in Arabidopsis. Plant Physiology 170, 686-701 (2016).
- Planchet, E., Jagadis Gupta, K., Sonoda, M. and Kaiser, W.M. Nitric oxide emission from tobacco leaves and cell suspensions: rate limiting factors and evidence for the involvement of mitochondrial electron transport. The Plant Journal 41, 732-743 (2005).
- Yamasaki, H., Sakihama, Y. and Takahashi, S. An alternative pathway for nitric oxide production in plants: new features of an old enzyme. Trends in Plant Science 4, 128-129 (1999).