Protein Nitration
Protein nitration involves the addition of a nitro group (–NO₂) to susceptible amino acids, mainly tyrosine (Tyr) and tryptophan (Trp), although most studies focus on tyrosine nitration. Nitration is generally considered irreversible and often impairs protein function, though neutral or positive effects have been reported. Importantly, tyrosine nitration is a selective, non-random process influenced by protein structure and cellular context (e.g., soluble vs. membrane-bound).
Mechanisms of Nitration
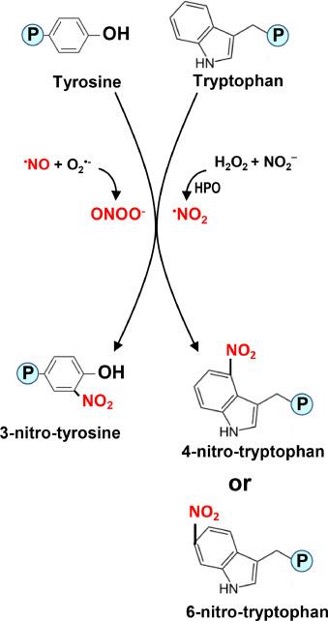
Tyrosine and tryptophan nitration mainly occur via peroxynitrite (ONOO⁻) or nitrogen dioxide radicals (•NO₂). Peroxynitrite is rapidly formed from nitric oxide (•NO) and superoxide (O₂•⁻) near sites of superoxide production and is highly reactive at physiological pH. Nitration proceeds indirectly through radical intermediates. Alternatively, •NO₂-mediated nitration can arise from reactions between hydrogen peroxide (H₂O₂) and nitrite (NO₂⁻) in the presence of hemoperoxidases.
Reversibility and Cellular Fate
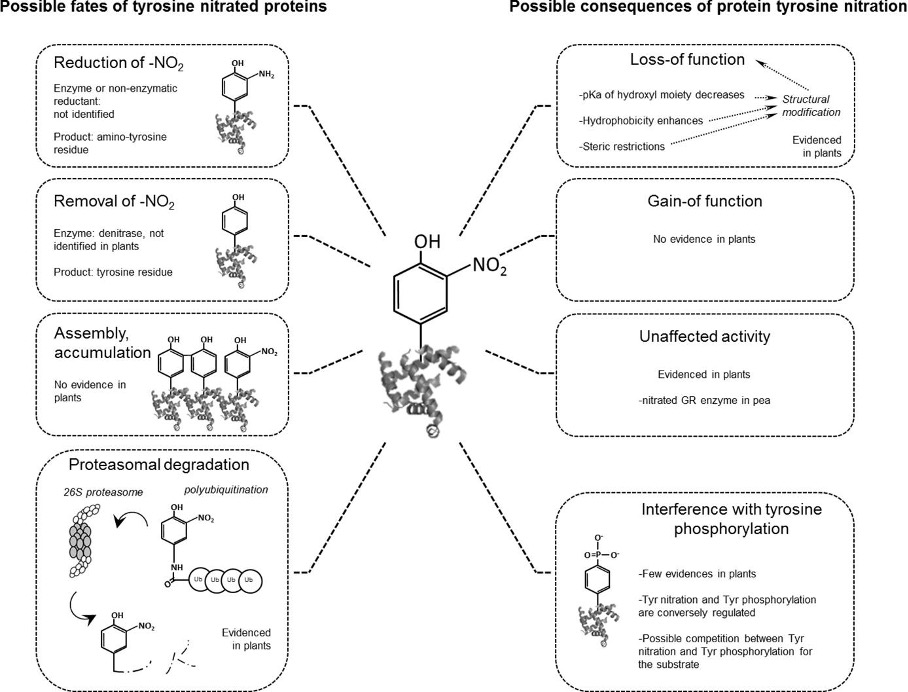
For tyrosine nitration to regulate signaling independently of phosphorylation, it must be reversible. Although once considered irreversible, denitration mechanisms have been identified in animals, while in plants no denitrase has yet been confirmed, leaving reversibility uncertain. Alternative outcomes include reduction of nitrotyrosine to aminotyrosine or removal of nitrated proteins via proteasome-mediated degradation. Evidence suggests that nitration often increases protein susceptibility to proteasomal degradation, including in plants.
Functional Effects and Phosphorylation
Functionally, protein tyrosine nitration lowers the pKa of tyrosine, increases hydrophobicity, and can induce structural and steric changes. In plants, nitration generally leads to loss of protein function, though exceptions exist. In animals, nitration can either activate, inactivate, or have no effect on protein activity, and functional changes are not always directly caused by tyrosine modification alone.
Tyrosine nitration can also positively or negatively affect tyrosine phosphorylation, thereby influencing cell signaling. While this relationship is well documented in non-plant systems, evidence in plants remains limited. Nonetheless, disruptions in microtubule organization and protein stability suggest a functional interplay between tyrosine nitration and phosphorylation, potentially through competition for the same tyrosine residues (Kolbert et al., 2017).
Explore Nitrated Proteins
Browse our comprehensive database of nitrated proteins with experimental evidence and predictions.
View Protein DatabaseReferences
- Corpas FJ, González-Gordo S, Palma JM Protein nitration: A connecting bridge between nitric oxide (NO) and plant stress.. Plant Stress 2, 100026 (2021). https://doi.org/10.1016/j.stress.2021.100026
- Kolbert Zs, Feigl G, Bordé Á, Molnár Á, Erdei L Protein tyrosine nitration in plants: Present knowledge, computational prediction and future perspectives.. Plant Physiology and Biochemistry 113, 56-63 (2017). https://doi.org/10.1016/j.plaphy.2017.01.028